A multisite, international phase 2 trial evaluating the investigational drug garetosmab has shown that it reduced soft-tissue flare-ups significantly and prevented new areas of abnormal bone formation in patients with fibrodysplasia ossificans progressiva (FOP).
Kathryn Dahir, MD, professor of Medicine in the Division of Endocrinology and Diabetes, served as the principal investigator at Vanderbilt University Medical Center for the randomized, double-blinded, placebo-controlled trial to study the efficacy and safety of garetosmab, a human monoclonal antibody. Garetosmab binds to activin A, a protein in the body related to growth, and blocks its ability to activate FOP-mutant ACVR1.
The findings, published this week in the journal Nature Medicine, give hope to individuals with the rare disease that effects 1 out of 1 million individuals in the United States. FOP is caused by mutations in the gene ACVR1 which lead to abnormal formation of bone outside of the normal skeleton in soft tissue including muscles, tendons and ligaments throughout the body.
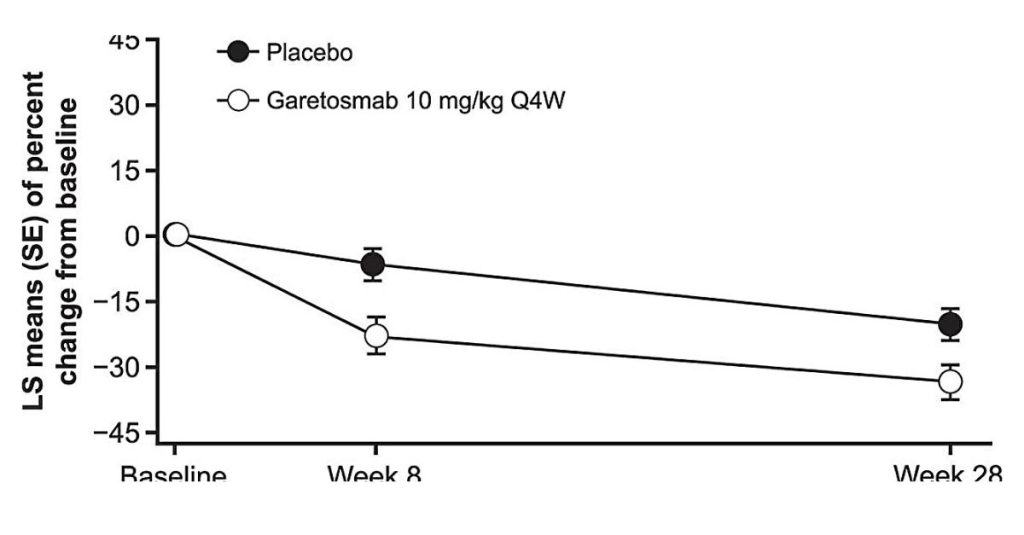
“Garetosmab decreased total lesion activity by nearly 25%, including new and existing lesions, as compared to placebo, and showed a remarkable 90% decrease in the number of new bone lesions. The prevention of new, heterotopic bone is of critical importance to the treatment of FOP,” said Dahir. “Patient-reported soft tissue flare-ups — with symptoms such as fever, pain, swelling and stiffness — were also reduced by half. As flare-ups often precede the formation of new lesions, we are encouraged that this investigational drug may help prevent the progression of FOP.
“The loss of mobility created by the abnormal bone growth is cumulative and steadily limits the ability of persons with FOP to perform even the most basic activities. It’s time for these patients and their families to feel hope with the possibility of a treatment that can make a real difference in their lives. I’m grateful to the patients and their families who participated in the trial, and we look forward to continuing to gather evidence on the safety and efficacy of this treatment.”
In the trial, 44 adults enrolled in the multicenter study who were diagnosed with FOP received garetosmab or a placebo every four weeks and were evaluated for new bone lesions by the using whole body positron emission tomography (PET) with sodium fluoride and low-dose computed tomography (CT) scans at baseline, eight weeks, 28 weeks and 56 weeks. At 28 weeks, all subjects began an open-label period in which both patients and researchers knew which treatment was being administered. Patients in the placebo group could choose to begin receiving garetosmab.
Brentwood, Tennessee, resident Sharon Kantanie, who was diagnosed with FOP at age 6, was one of the participants in the drug trial at VUMC. When the study shifted to the open-label period she learned she had received garetosmab from the beginning. Kantanie primarily experienced minor dermatological side effects. She is happy the results have been published and that further investigation of the drug’s efficacy and safety is now in progress.
“There are really no words to adequately explain FOP’s effects on a person and the disease’s impact on their family and caregivers,” Kantanie said. “To move closer to an effective treatment is a wonderful thing. And to have that treatment provided earlier in life would be amazing so people with FOP and their families won’t have to go through the extremes and painful progression of this disease as we’ve experienced.”
There were five deaths during the trial — a relatively high number for a small study. After a pause to investigate, the deaths were determined to be unlikely related to the drug intervention. It was concluded that the deaths were most likely associated with the severity of the participant’s baseline disease or to preexisting co-morbidities and were consistent with known causes of death and life expectancy for patients with FOP.
Researchers from 11 institutions across the United States, as well as Italy, England, France, Poland, Spain, Canada and the Netherlands participated in the study. Garetosmab was developed by Regeneron Pharmaceuticals which funded the trial.
Dahir is currently one of three U.S. principal investigators in a phase 3 adult Optima trial to further assess the benefits and risks of garetosmab for individuals with FOP and to validate the phase 2 results. A pediatric trial is also planned.
The research at VUMC is supported by the National Institutes of Health (TR002243).
Source: Science Daily